(8) Ustekinumab for Crohn’s disease: A nationwide long-term real-life observational cohort study on Finnish patients
Författare/Medförfattare
Clas-Göran af Björkesten [1], Tuire Ilus [2], Taru Hallinen [3], Erkki Soini[3], Anja Eberl[1], Kalle Hakala[4], Mikko Heikura[5], Eija Hirsi[6], Airi Jussila[2], Mikko Kellokumpu[7], Ritva Koskela[8], Inka Koskinen[9], Veikko Moilanen[10], Christian Nielsen[11], Urpo Nieminen[1], Heikki Nuutinen[12], Markku Heikkinen[13], Ulla-Maija Suhonen[14], Jyrki Tillonen[15], Karri Utriainen[16], Ilkka Vihriälä[17], Christina Wennerström[18, 19], Andras Borsi[20], Minni Koivunen[21], Taina Sipponen[1]
Affiliates
University of Helsinki and Helsinki University Hospital, Helsinki, Finland[1], Tampere University Hospital, Tampere, Finland[2], ESiOR Oy, Kuopio, Finland[3], Kanta-Häme Central Hospital, Hämeenlinna, Finland[4], North Karelia Central Hospital, Joensuu], Finland[5], South Karelia Central Hospital, Lappeenranta, Finland[6], Lapland Central Hospital, Rovaniemi, Finland[7], Oulu University Hospital, Oulu, Finland[8], Central Finland Central Hospital, Jyväskylä], Finland[9], Satakunta Central Hospital, Pori], Finland[10], Vaasa Central Hospital, Vaasa, Finland[11], Turku University Hospital, Turku, Finland[12], Kuopio University Hospital, Kuopio, Finland[13], Kainuu Central Hospital, Kajaani, Finland[14], Päijät-¬Häme Central Hospital, Lahti, Finland[15], Turku University Hospital, Salo Hospital, Salo, Finland [16], Central Ostrobothnia Central Hospital, Kokkola, Finland[17], Medical Affairs, Janssen Cilag AB, Solna, Sweden[18], Department of Epidemiology Research, Statens Serum Institut, Denmark, Copenhagen[19], Janssen Cilag Limited, EMEA HEMAR, High Wycombe, United Kingdom[20], Medical Affairs, Janssen Cilag Oy, Espoo, Finland[21].
Abstract
Background
Real-life long-term evidence on ustekinumab treatment in patients diagnosed with Crohn’s Disease (CD) is limited. The present study is a retrospective non-interventional nation-wide chart review of dosing and long-term clinical outcomes in Finnish CD patients treated with ustekinumab (FINUSTE2, EUPAS30920).
Methods
FINUSTE2 involved 17 Finnish hospitals. Eligible patients were adults with CD, initiating ustekinumab therapy with intravenous induction during 2017 and 2018. Disease activity data, such as faecal calprotectin (fCal), the modified (abdominal palpation finding omitted) Harvey Bradshaw index (mHBI) and the Simple Endoscopic Score for Crohn’s disease (SES-CD) were collected, among other defined time points, at 16 weeks and one year. An mHBI of 4 points or less or an mHBI reduction of at least 3 points from baseline was defined as clinical benefit.
Results
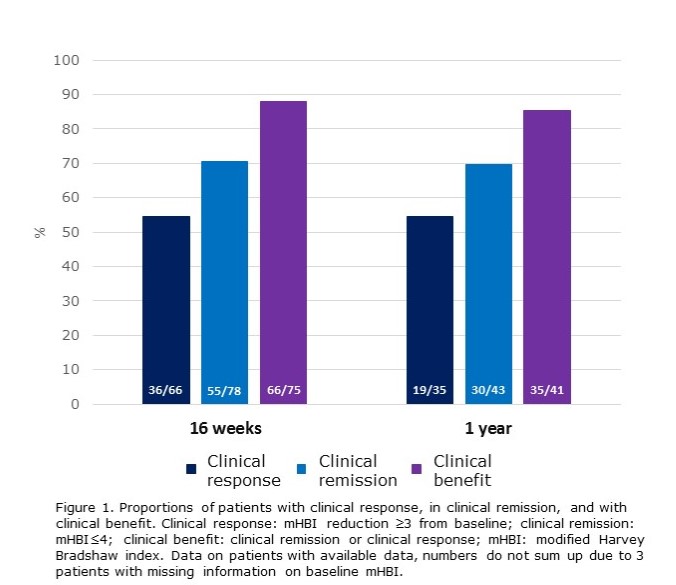
155 patients (48% female) with a median age of 41 and disease duration of 14 years initiated ustekinumab treatment for CD. The disease was stricturing in 54% and penetrating in 15% of patients, 59% had prior CD-related surgeries, 30% had a history of one and 66% had a history of two or more biologic agents. After induction, 140 patients (90.3%) continued to maintenance treatment. Among the 93 patients with one-year follow-up, 77 were still on ustekinumab. Among ustekinumab treated, SES-CD (median at baseline 10, at one year 3, p=0.033) and fCal (776 μg/g at baseline, 305 μg/g at one year, medians, p<0.001) decreased significantly in those patients with data available. Of those still on ustekinumab treatment at one year, 85% showed clinical benefit, and the proportion of patients on corticosteroids decreased from 41% at baseline to 13% at one year (p<0.001). Figure 1 describes clinical outcomes at 16 weeks and one year among patients with available data.
Conclusion
In this nation-wide real-life study, covering all major centres in Finland, ustekinumab treatment of patients with highly refractory and long-standing CD effectively reduced inflammatory activity, induced long-term benefit, and allowed for significant corticosteroid tapering.