(7) SERUM N-GLYCOMIC BIOMARKERS PREDICT TREATMENT ESCALATION IN INFLAMMATORY BOWEL DISEASE
Författare/Medförfattare
Archana Shubhakar (1,2), Bas C Jansen(1), Alex T. Adams(3), Karli R. Reiding(4,5), Nicholas T. Ventham(6), Rahul Kalla(6), Daniel Bergemalm(7), Paulina A Urbanowicz(1), Richard A Gardner(1), IBD-BIOM Consortium, Jonas Halfvarson(7)*,Jack Satsangi(3), Daryl L Fernandes(1), Manfred Wuhrer(4), Daniel I R Spencer(1) *Presenting author
Affiliates
(1) Ludger Ltd, Culham Science Centre, Abingdon, Oxfordshire, United Kingdom, (2) Division of BioAnalytical Chemistry, VU University Amsterdam, Amsterdam, The Netherlands, (3) Translational Gastroenterology Unit, University of Oxford, Oxford, United Kingdom, (4) Center for Proteomics and Metabolomics, Leiden University Medical Center (LUMC), Leiden, The Netherlands, (5) Division of Biomolecular Mass Spectrometry and Proteomics, Utrecht University, Utrecht, The Netherlands, (6) Institute of Genetics and Molecular Medicine, University of Edinburgh, Edinburgh, United Kingdom, (7) Department of Gastroenterology, Faculty of Medicine and Health, Örebro University, Örebro, Sweden
Abstract
Background & Aims:
The clinical management of Inflammatory Bowel Disease (IBD) would benefit from biomarkers allowing treatment choices to be optimised for each individual patient. In this study we have investigated and validated composite N-glycomic biomarkers in predicting response to primary treatment following diagnosis.
Methods:
Total N-glycans from 227 patients and 195 controls (Edinburgh, UK) were assessed in a 10ul serum sample collected at the index clinic visit and analyzed by automated high-throughput fluorescent labeling of glycans using ultra-high performance liquid chromatography. Multivariable logistic regression was performed on 24 derived glycomics traits, based on structurally related glycoforms. We created cox proportional hazard models to characterize biomarkers to predict the need for treatment escalation – defined as a requirement for anti-TNF, biologics, or surgery. An independent cohort of 49 patients (15 requiring treatment escalation) was recruited in Orebro (Sweden) for validating the escalation biomarker.
Results:
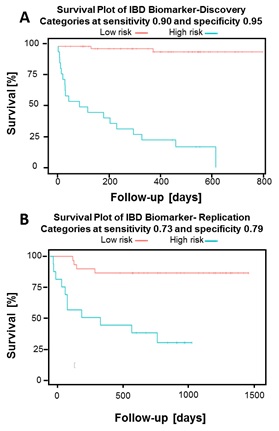
The requirement for escalation of treatment in newly diagnosed IBD cases could be predicted using a panel of 7 derived glycan traits (Figure 1A, HR = 25.91 and p = 1.12 ×10-12), with similar panels of markers for CD and UC separately. This model also predicted the need for treatment escalation in the replication cohort (Figure 1B, HR = 5.07 and p = 1.14 ×10-5). Additionally, we have described alterations in glycosylation seen in IBD, and the correlations between glycan traits and various clinical factors and common laboratory investigations.
Conclusions:
These results demonstrate that measurement of total serum N-glycans in new IBD patients can provide insight into future response to treatment. In the future these biomarkers like these could prove valuable in personalizing initial treatment of IBD.